Nivolumab
CheckMate 017 and 057
29
LCSS = Lung Cancer Symptom Scale; ORR = objective response rate; OS = overall survival; PD = progressive disease;
PFS = progression-free survival; TKI = tyrosine kinase inhibitor
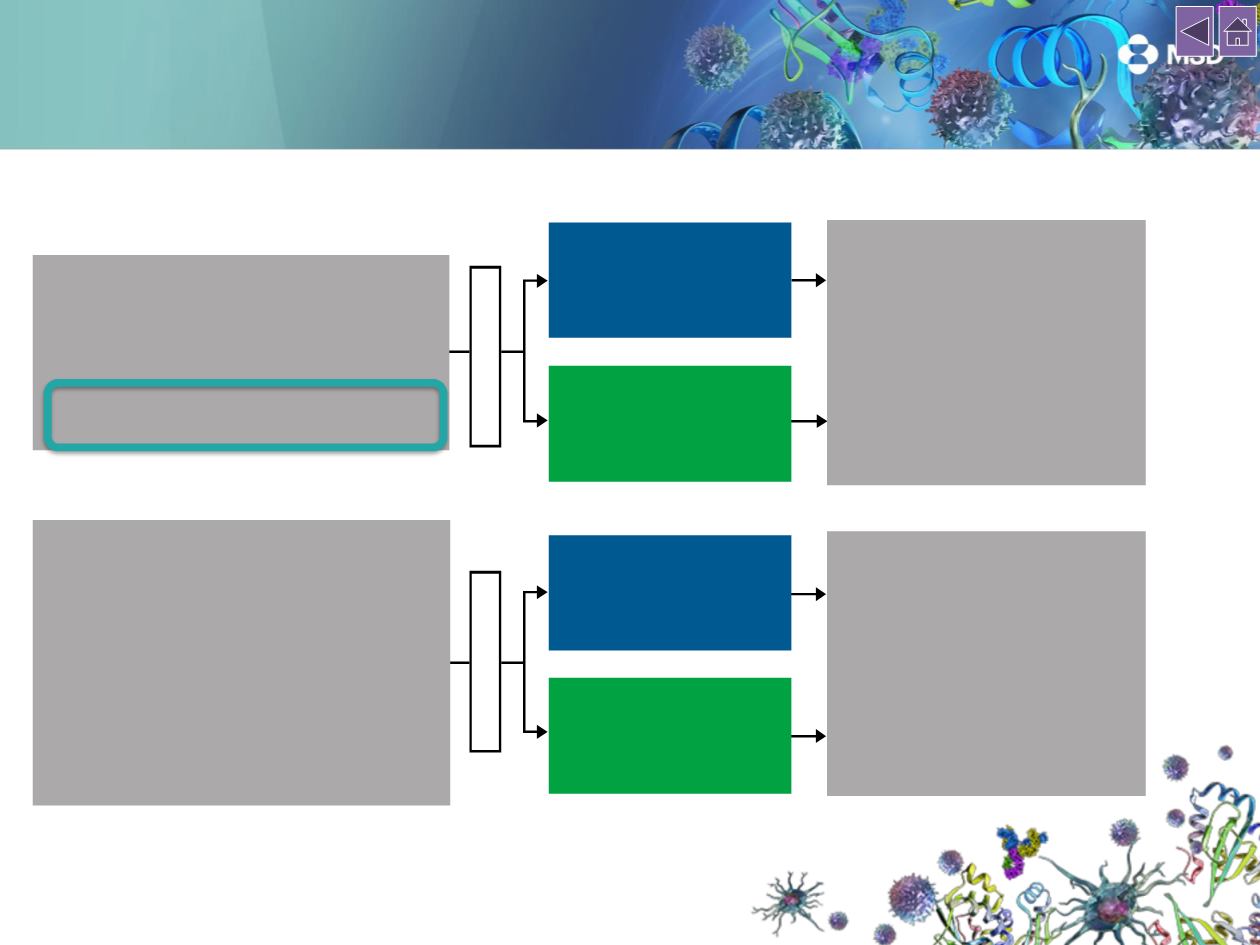
CheckMate 017
(NCT01642004; N = 272)
CheckMate 057
(NCT01673867; N = 582)
Docetaxel 75 mg/m2 IV Q3W
until PD or
unacceptable toxicity
(n = 290)
Key eligibility criteria
• Stage IIIB/IV
SQ NSCLC
• ECOG PS 0–1
• One prior platinum-based chemotherapy
•
Pretreatment (archival or fresh) tumor
samples required for PD-L1 analysis
Key eligibility criteria
• Stage IIIB/IV
non-SQ NSCLC
• ECOG PS 0–1
• One prior platinum-based chemotherapy
•
Pretreatment (archival or fresh) tumor
samples required for PD-L1 analysis
• Prior maintenance therapy allowed
• Prior TKI therapy allowed for known
ALK
translocation or
EGFR
mutation
Randomize 1:1
Randomize 1:1
Endpoints
•
Primary
‒
OS
•
Additional
‒
ORR
‒
PFS
‒
Efficacy by tumor PD-L1
expression
‒
Safety
‒
Quality of life (LCSS)
Endpoints
•
Primary
‒
OS
•
Additional
‒
ORR
‒
PFS
‒
Efficacy by tumor PD-L1
expression
‒
Safety
‒
Quality of life (LCSS)
Nivolumab 3 mg/kg IV Q2W
until PD or
unacceptable toxicity
(n = 292)
Docetaxel 75 mg/m2 IV Q3W
until PD or
unacceptable toxicity
(n = 137)
Nivolumab 3 mg/kg IV Q2W
until PD or
unacceptable toxicity
(n = 135)
Brahmer J et al. N Engl J Med 2015
Borghaei H, et al. N Engl J Med 2015


