First line ongoing trials
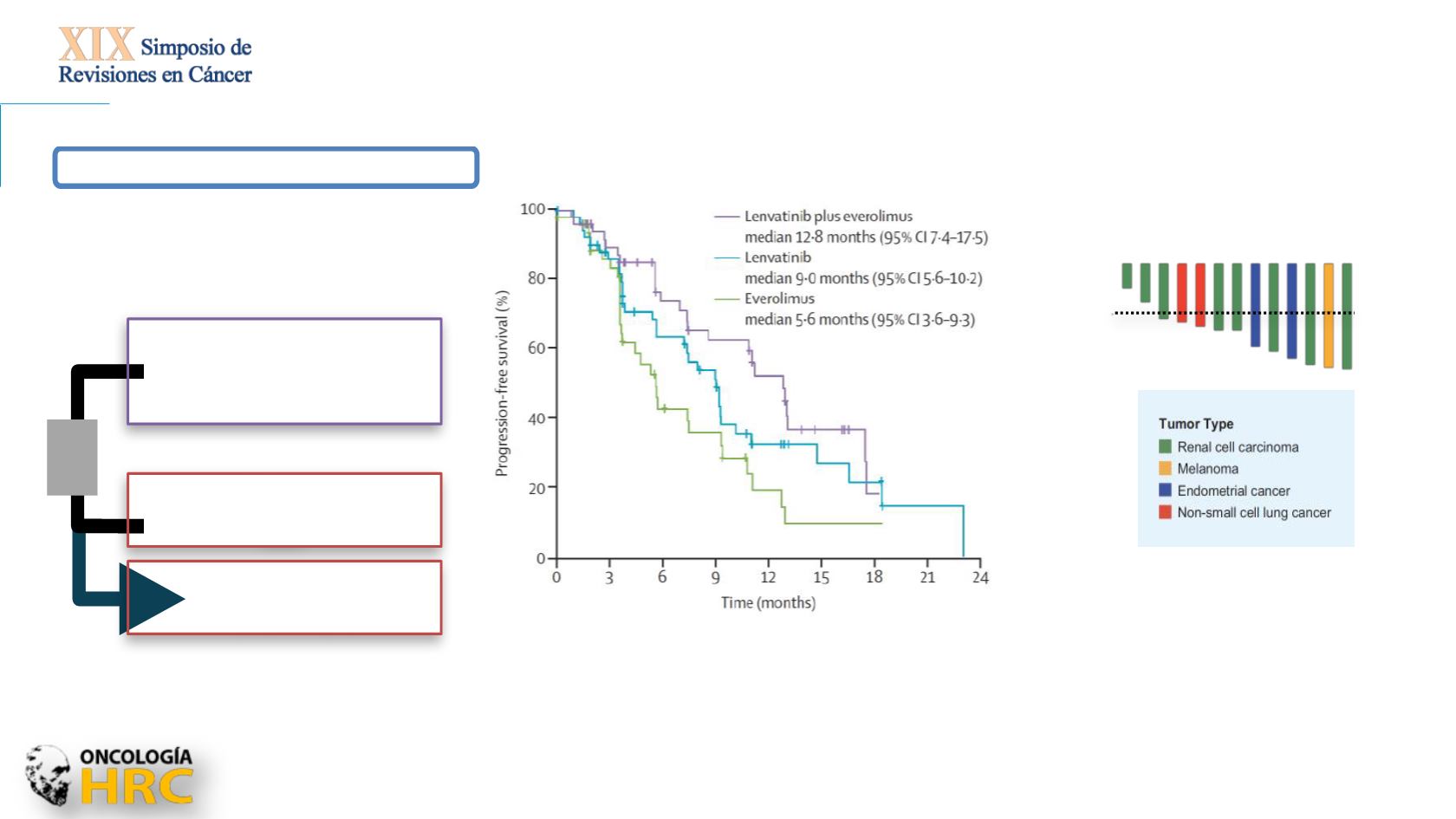
Motzer RJ, et al. Lancet Oncol 2016
Taylor MH, et al. ESMO 2016
CLEAR
Phase III
Lenvatinib + pembro
20 mg/day + 200 mg (IV) every 3
weeks
Sunitinib
50 mg/day 4/2
Primary endpoint: PFS
Combination VEGFR + mTOR/PD-1 inhibition
R
n=735
Lenvatinib + everolimus
18 mg/day + 5 mg/day


