First line trials: closed for recruitment
McDermott DF, et al. J Clin Oncol 2016
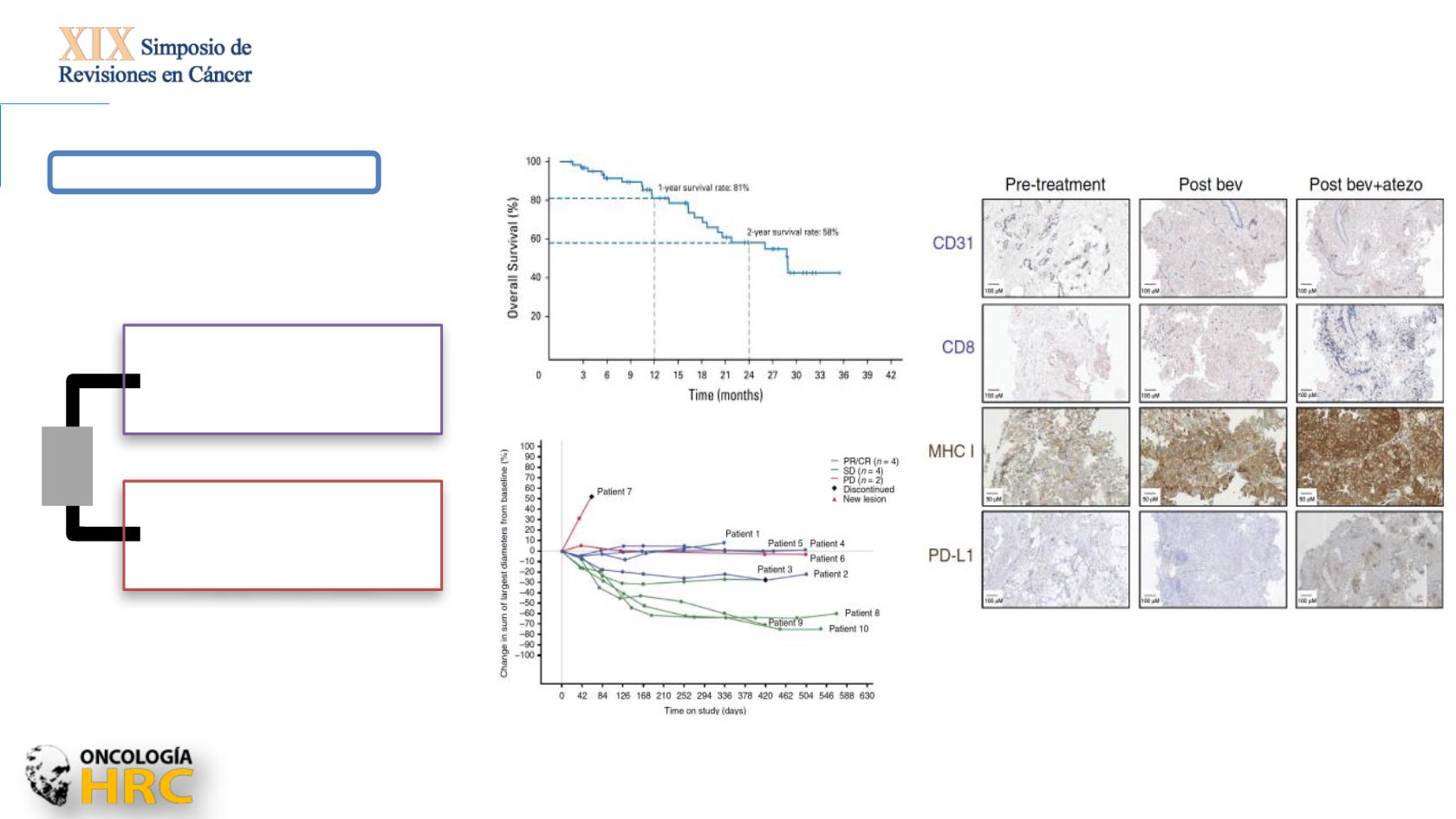
Wallin JJ, et al. Nat Commun 2016
IMmotion151
Phase III
Atezolizumab +
bevacizumab
1200 mg IV +
15 mg/kg IV every 3 weeks
Sunitinib
50 mg/day 4/2
Co-Primary endpoint: PFS, OS
PD-L1 + VEGF inhibition
R
n=830


