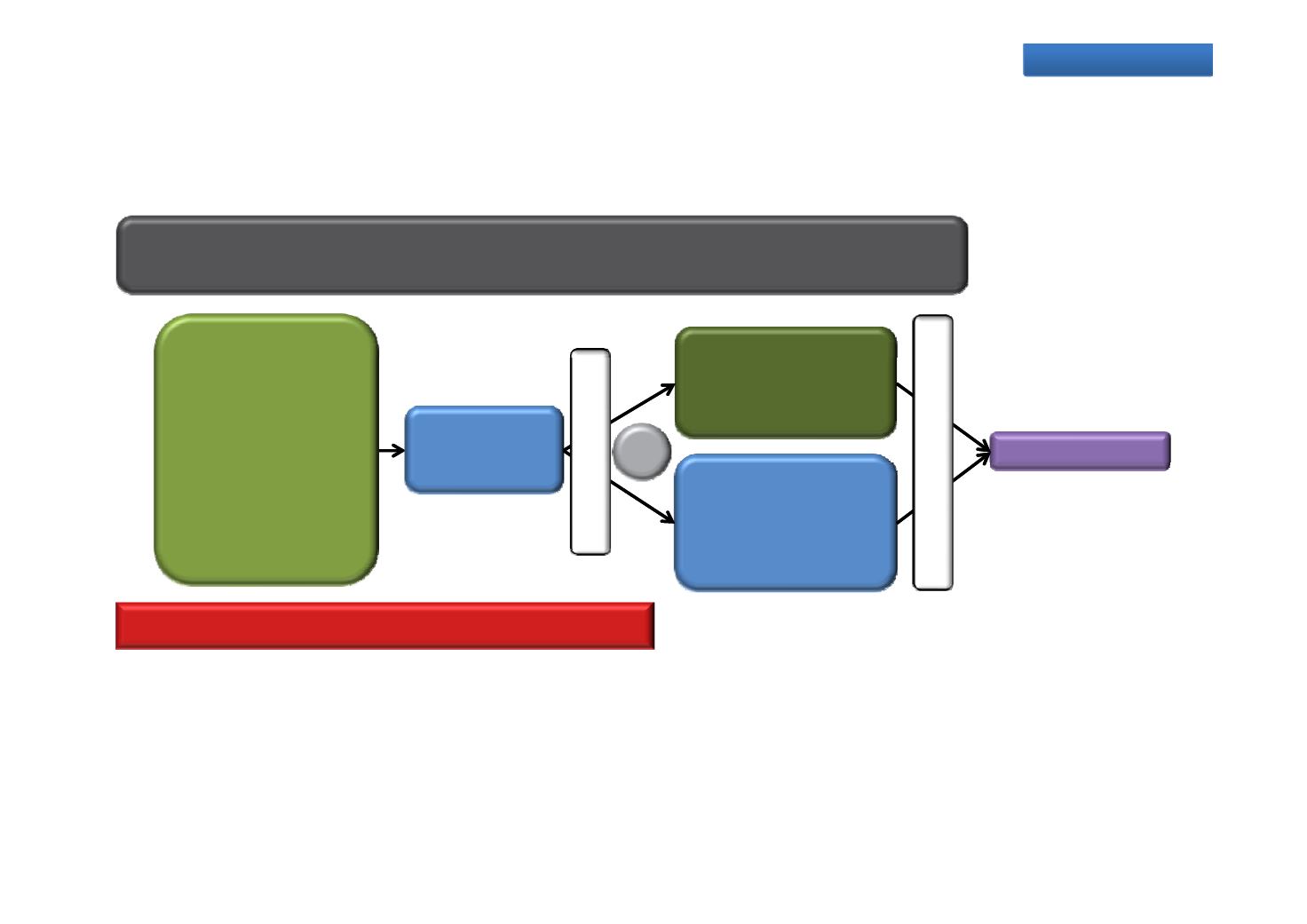
PLATO: Continued enzalutamide treatment
beyond progression in combination with
Ongoing trials
abiraterone in chemotherapy-naïve mCRPC
•
A Phase 4 randomised, double‐blind, placebo‐controlled study
•
Primary endpoint: PFS
n=509
Placebo (QD) +
Chemotherapy‐
naïve mCRPC after
ADT failure
ECOG PS 0 1
Open‐label
enzalutamide
Safety follow‐up
abiraterone
(1000 mg QD) +
prednisone (5 mg BID)
PS
A
R
R
1:1
PR
O
GR
ES
S
–
Estimated life
expectancy
≥12 months
160 mg QD
Enzalutamide
(160 mg QD) +
abiraterone (1000 mg
QD) + prednisone
(5 mg BID)
I
SE
I
O
N
Enrolment complete
Planned evaluations
•
PFS, time to PSA progression, PSA response, safety
Timing
•
Estimated study completion December 2016
ADT=androgen‐deprivation therapy; BID=twice daily; ECOG PS=Eastern Cooperative Oncology Group performance status; mCRPC=metastatic castration‐
resistant prostate cancer; PFS=progression‐free survival; PSA=prostate‐specific antigen; QD=once daily; R=randomisation.
NCT01995513. Available at
. Last accessed: May 2015.
2
0


