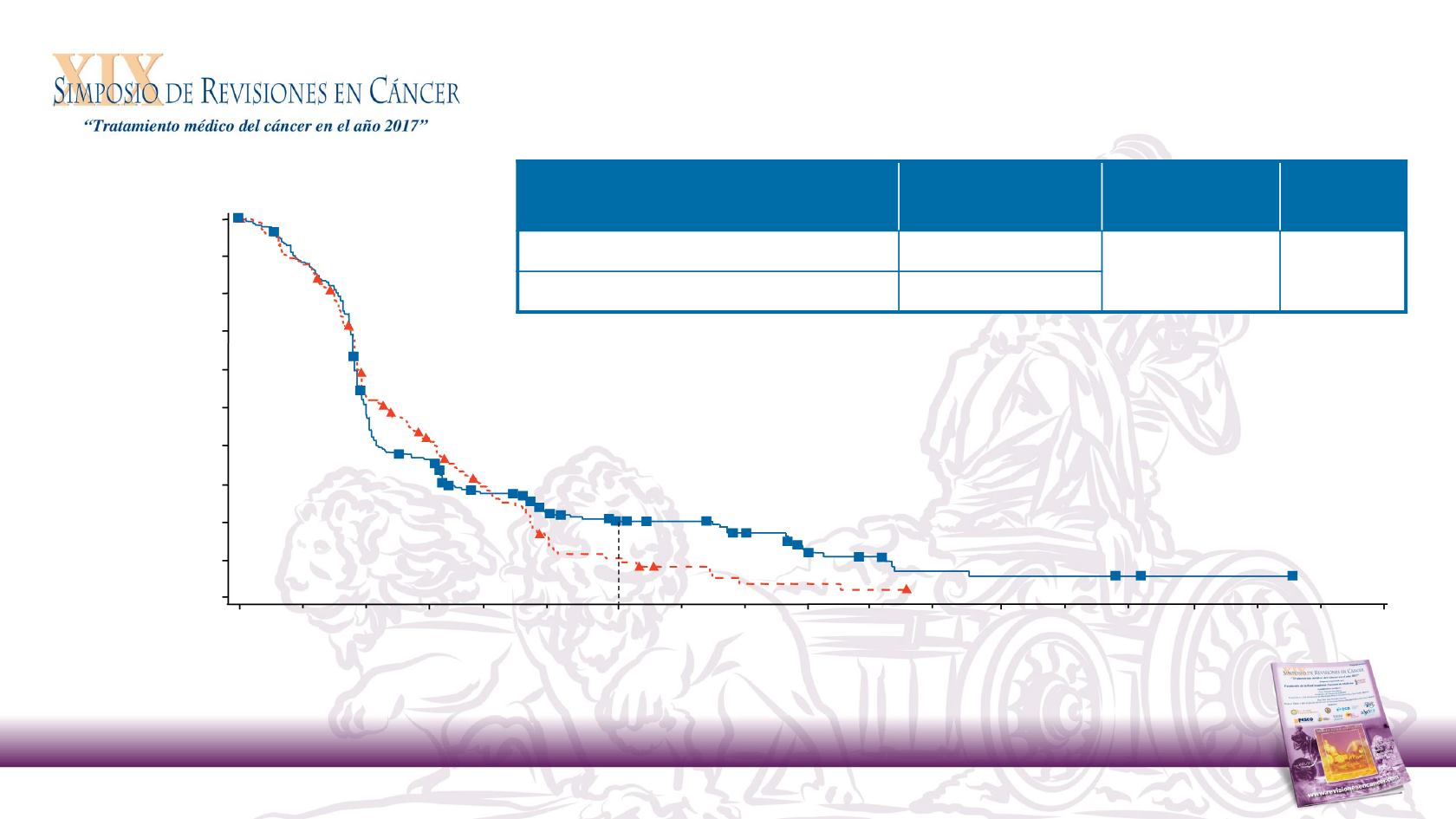
Progression-Free Survival
Nivolumab in R/M SCCHN After Platinum Therapy
25
Months
Nivolumab
240
79
32
12
4
1
0
Investigator’s
Choice
121
43
9
2
0
0
No. at Risk
0
Progression-Free Survival
(% of patients)
0
3
6
9
12
15
18
0
10
20
30
40
50
60
70
80
90
100
6-month PFS rate (95% CI)
19.7%
(14.6, 25.4)
9.9%
(5.0, 16.9)
Median OS, mo
(95% CI)
HR
(97.73% CI)
P
-value
Nivolumab (n = 240)
2.0 (1.9, 2.1)
0.89
(0.70, 1.1)
0.3236
Investigator’s Choice (n = 121)
2.3 (1.9, 3.1)


